ALL MACHINES
Complete Product Range
Filter by application category or browse the full portfolio.

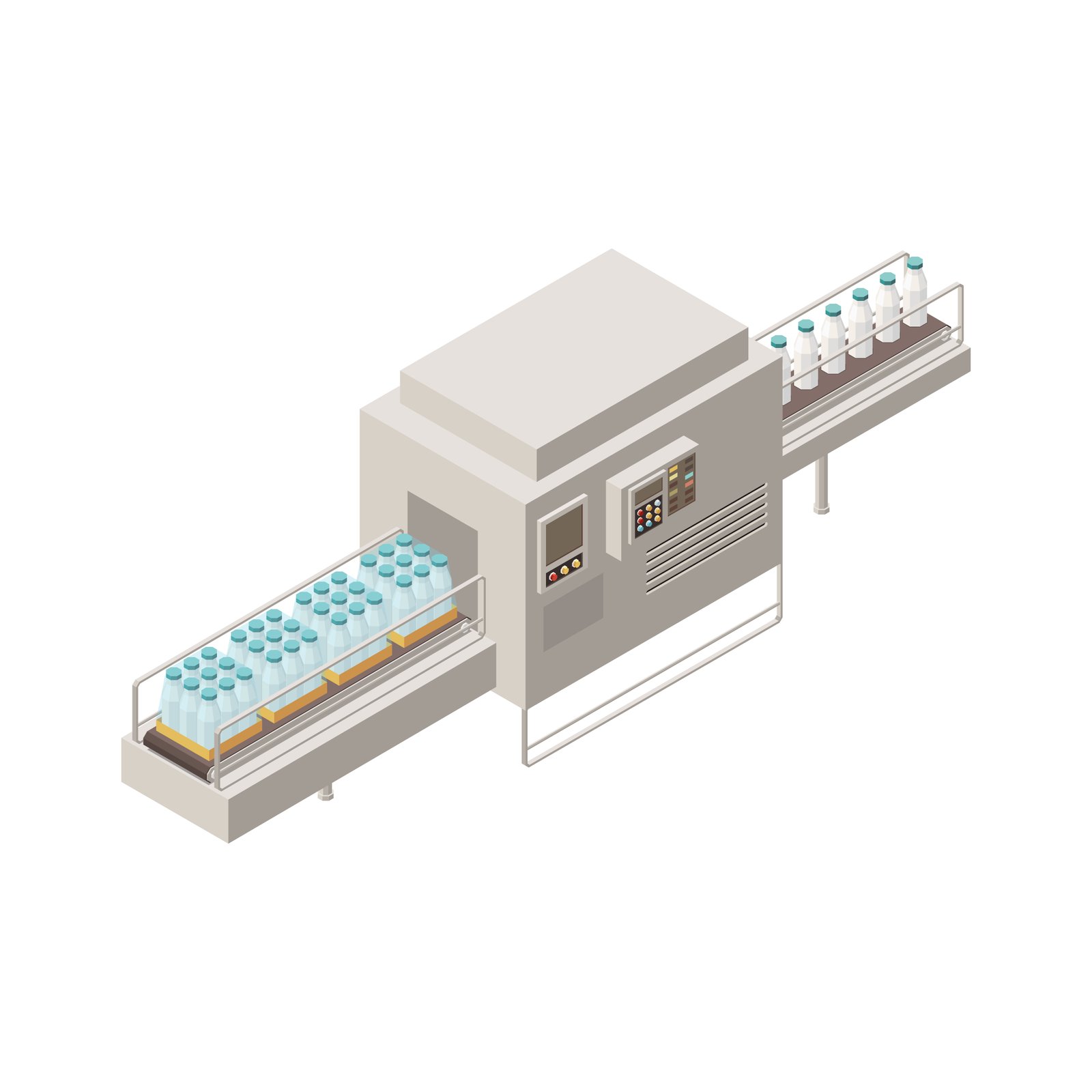




PRODUCT CATALOGUE
From aseptic vial filling to intelligent inspection — every machine engineered to GMP specification, validated, and documented for global regulatory markets.
ALL MACHINES
Filter by application category or browse the full portfolio.






MACHINE COMPARISON
* All specifications subject to configuration. Contact our engineers for your specific requirements.
APPLICATIONS
Deep application knowledge for every pharmaceutical manufacturing challenge.
INJECTABLE & PARENTERALS
Complete aseptic filling lines with integrated isolator systems, RABS, or conventional barrier technology. Meeting FDA 21 CFR, EU GMP Annex 1, and PMDA requirements for sterile injectable products.
ORAL SOLID DOSE
High-speed rotary tablet presses, capsule filling machines, and sachet lines. Full compliance with EU GMP, WHO GMP, and ICH Q7 for generic and branded solid dose products.
BIOLOGICS & ASEPTIC
Specialized processing for biologic and biosimilar products including lyophilization, cold-chain compatible filling, and single-use technology integration. Compliant with EU GMP Annex 2.
SECONDARY PACKAGING
Complete secondary packaging solutions from blister forming to carton erection and serialization. Fully integrated with track & trace and GS1 compliance for EU FMD and US DSCSA.
WHO WE SERVE
High-volume, validated production lines with full SCADA integration, real-time OEE monitoring, and global regulatory compliance.
Flexible, small-batch systems for mAbs, cell therapies, and gene therapies — compatible with single-use technology and aseptic processing.
Multi-product, quick-changeover platforms with comprehensive validation packages. Serving contract manufacturers across multiple customers and formats.
Cost-efficient, high-speed platforms built for WHO GMP and ICH Q7 compliance. Proven in emerging markets across Asia, South America, and the Middle East.
Assembly, packaging, and inspection systems for combination products, drug-device combinations, and ISO 13485-compliant device manufacturing.
Our application engineers will analyze your product, process, and regulatory requirements — and recommend the right platform for your facility.
Speak to an Engineer →